High-resolution separation of complex protein mixtures
Sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE) is frequently used to obtain high-resolution separation of complex protein mixtures. However, poor band separation can occur due to issues at multiple steps, such as improper sample preparation or insufficient buffering capacity during gel electrophoresis. Complete lysis, improving protein solubility, and quality buffers can help deliver the clearly-resolved, straight bands you need for analysis and downstream applications.
Consult with Boston BioProducts to get high-resolution separation on your gels.
The Process
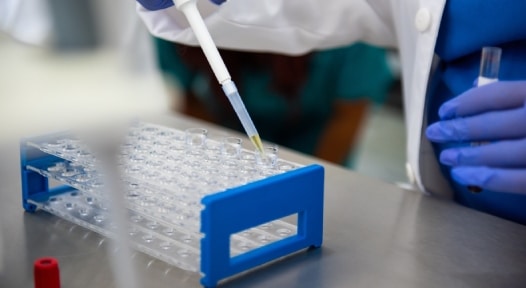
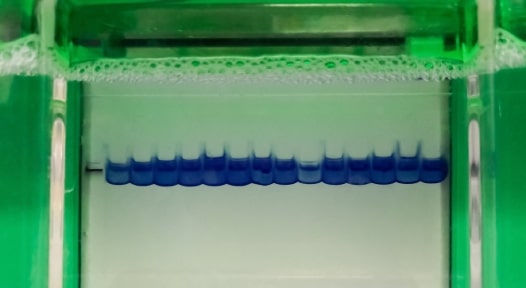
Lysed tissues or cells are denatured in SDS buffer containing loading dye. Once gels are cast, prepared samples are loaded onto the gel and then separated by size via polacrylamide gel electrophoresis (PAGE). Following resolution, proteins can be visualized by staining with Coomassie, silver, or fluorescent stains.
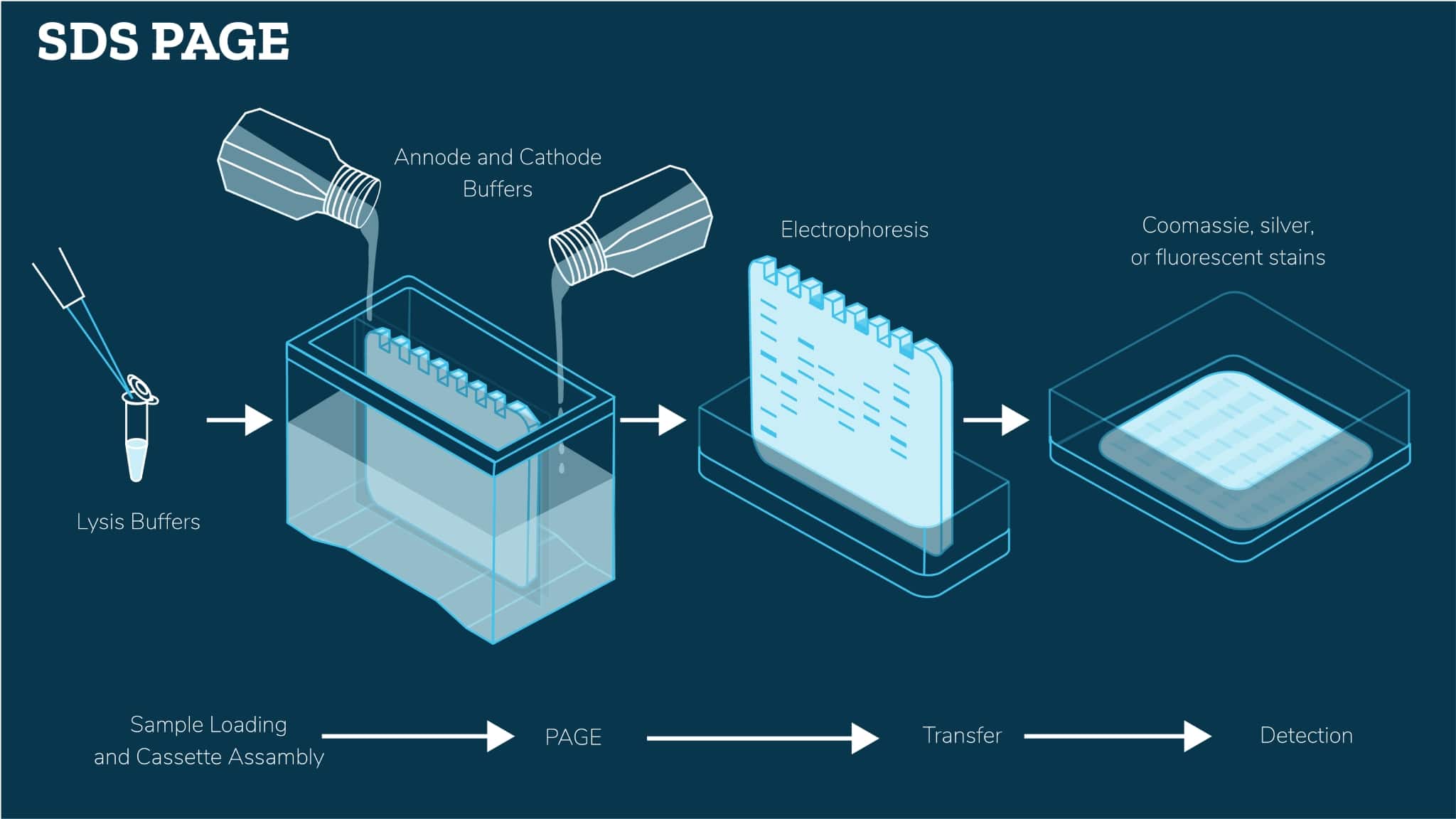
How we simplify the process
Most often when our customers contact us regarding difficulty in obtaining high-resolution separation of complex mixtures of proteins via SDS-PAGE, we look at optimizing the following parameters:
Lysis Buffer
It is essential that the samples (e.g., cells, tissues) are efficiently lysed. Identifying the right lysis buffer with the appropriate detergents for your protein(s) of interest is important. For example, membrane proteins require more thorough lysis to extract them from membranes.
SDS Sample Buffer
SDS in the sample buffer denatures proteins and gives them a negative charge to facilitate protein resolution during gel electrophoresis. Incomplete denaturation may lead to blurry bands, while insufficient negative charge will alter protein mobility.
Gel Casting Buffer
The pH of gel casting buffers will significantly influence SDS-PAGE protein resolution. Tris-Glycine gels are basic (pH 8.6) and offer lower resolution compared to acidic Bis-Tris gels (pH 6.4). Tris-acetate gels, with a pH of 7, are best for very large proteins. The selection of running buffer depends on gel composition and the proteins of interest.
Staining Solutions
Visualizing resolved proteins, especially at low abundance, requires a sensitive staining method. Fluorescent stains will generally be the most sensitive.
Ways Boston BioProducts can help
Learn more about how
Boston BioProducts can help
Featured Products for SDS PAGE
Featured Lysis Buffers
Featured Gel Casting Buffers and Solutions
Featured Sample Buffers
Featured Electrophoresis Buffers
Featured Services & Resources

Quality Assurance
We vet reliable distributors, have a proven quality management system, and in-house manufacturing to ensure consistent high-quality products.

Certificate Request
Transparency is central to our approach to customer service and we want to ensure that you can trust your reagents.

Order Support
With a simple, reliable, and flexible ordering process, we prioritize getting your SDS PAGE reagents to you quickly and securely.